Overview
The study applies single-particle cryo-electron microscopy to characterize the conformational ensemble of full-length, fully active rat cytochrome P450 reductase (CPR) in solution. The work addresses the unresolved relationship between available crystal structures and the solution-state conformations that underlie CPR-mediated interflavin and cytochrome electron transfer. A 3.3 Å reconstruction of the 77 kDa enzyme is reported, representing the first electron microscopy structure for this size class and enabling comparison between compact solution conformers and extant crystallographic models.
Methods and approach
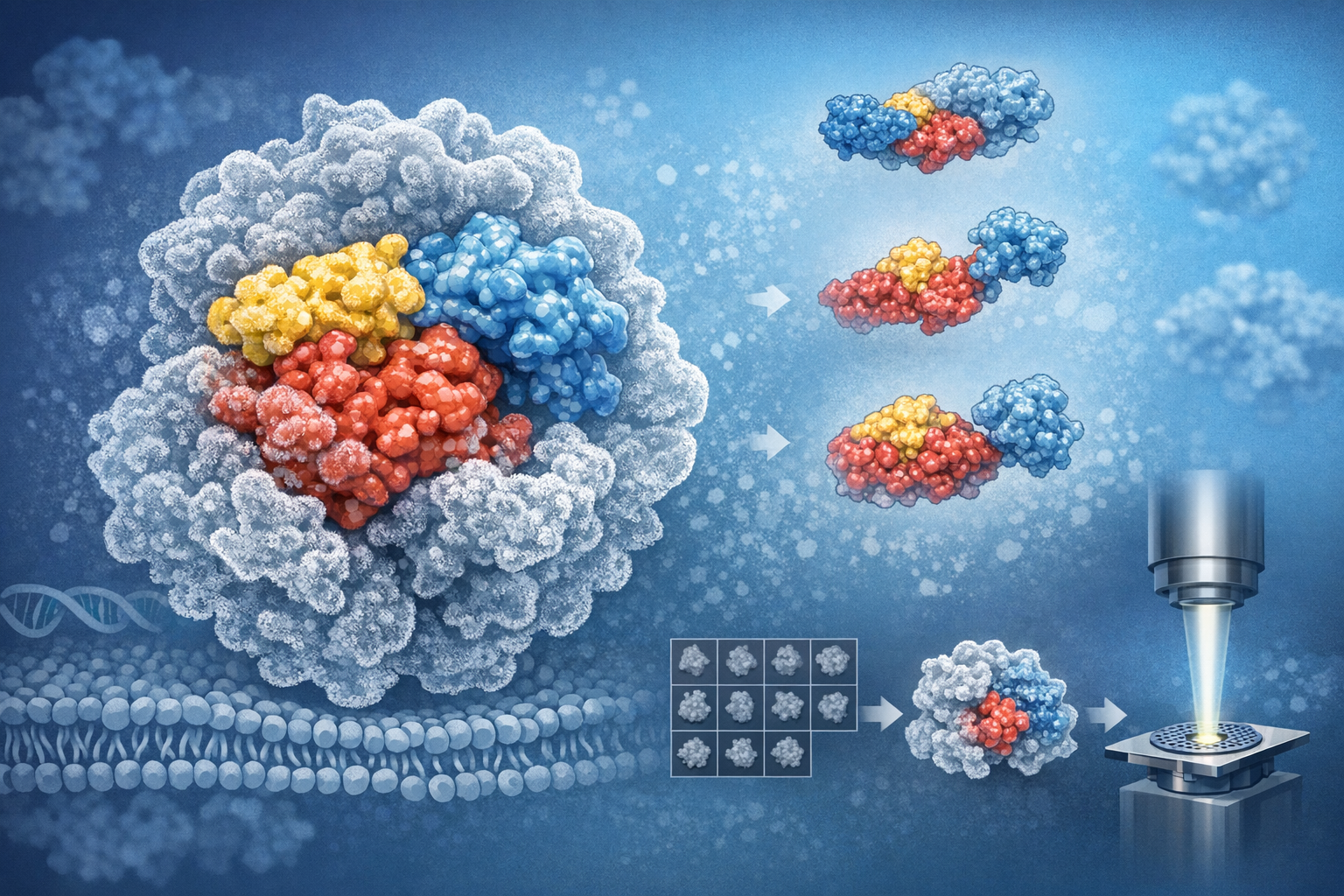
Recombinant full-length rat CPR was prepared in a state retaining catalytic activity and vitrified for single-particle cryo-EM. Data processing included particle selection, 2D classification, 3D refinement to a 3.3 Å consensus map, and focused 3D classification to resolve conformational heterogeneity of individual domains. Map interpretation leveraged existing high-resolution domain structures to place FMN-, FAD-, and connecting linker regions; population fractions were estimated from classification statistics and particle assignments.
Results
The dominant population adopts a compact interdomain arrangement consistent with an electron-transfer competent architecture, but the compact solution conformation is less tightly packed than previously reported crystal structures, indicating increased interdomain separation or flexibility. Focused classification revealed structurally distinct, less-populated variants of compact conformers. Approximately 20% of particles were assigned to classes in which the FMN-binding domain was either not resolved in the density or was displaced to positions remote from the catalytic FAD/core, indicative of large-amplitude domain displacement or high mobility.
Implications
These structural observations corroborate models in which large-scale interdomain rearrangements constitute the mechanistic basis for CPR function, coupling dynamic domain repositioning to sequential electron transfer events. The presence of a substantial minority population with an unresolved or displaced FMN domain emphasizes the importance of ensemble behavior for interpreting catalytic cycles and electron transfer gating. Methodologically, the study demonstrates that high-resolution cryo-EM combined with focused classification can access conformational subpopulations in small, flexible redox enzymes, informing mechanistic hypotheses that are not fully captured by static crystal structures.
Disclosure
- Research title: Cryo‐ EM reveals an ensemble of cytochrome P450 reductase conformations in solution
- Authors: Galina I. Lepesheva, Tatiana Y. Hargrove, Yi Ren
- Publication date: 2026-01-21
- DOI: https://doi.org/10.1002/pro.70448
- OpenAlex record: View
- Disclosure: This post is an AI-generated summary of a research work. It was prepared by an editor. The original authors did not write or review this post.