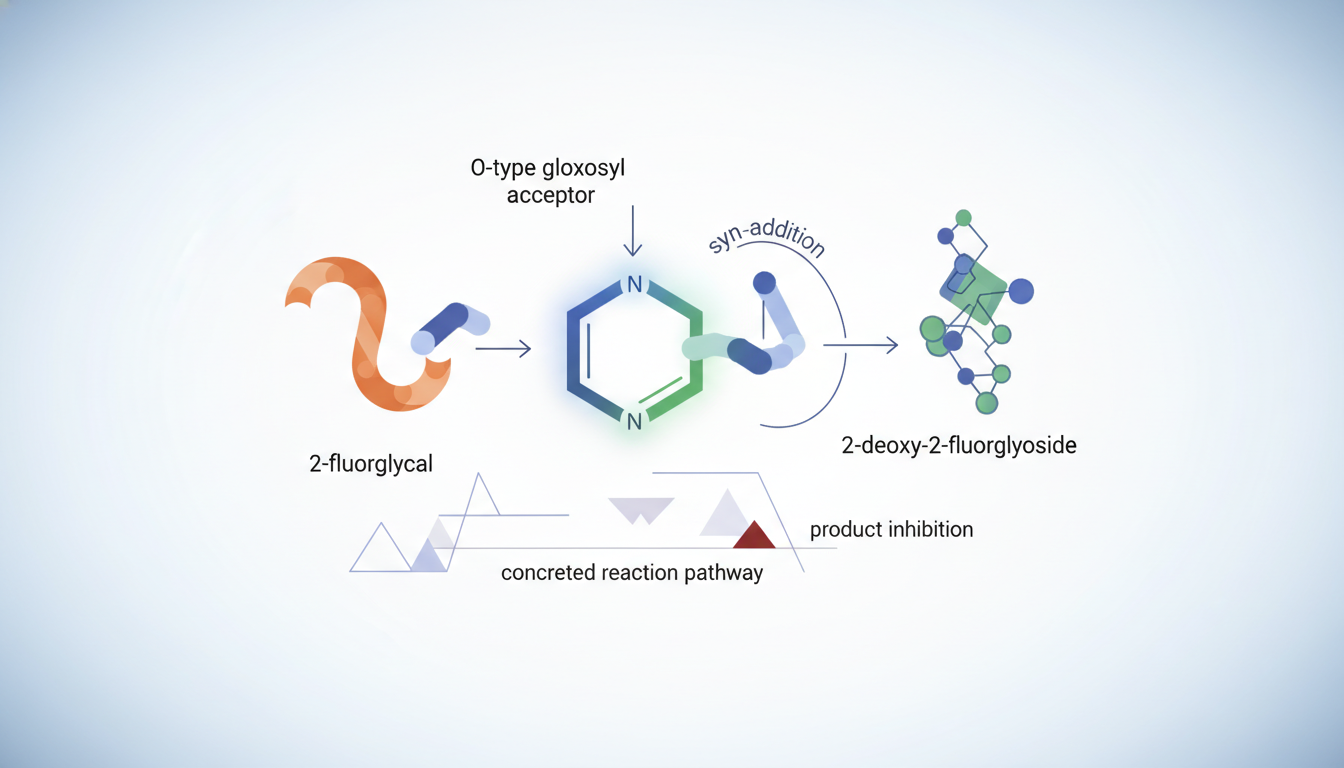
Chemists developed a pyridinium-salt-catalyzed method to make 2-deoxy-2-fluoroglycosides with high control over the arrangement of atoms in the product. The reaction adds an O-type glycosyl acceptor to 2-fluoroglycals and gives a structurally varied set of 2-fluoroglycosides. The outcomes strongly favor syn-addition, and experiments with deuterium support a concerted reaction pathway. Additional experiments and calculations revealed that product inhibition occurs and help explain lower efficiency with electron-poor phenols and with alcohol substrates.
What the study examined
This work explored a chemical route to make 2-deoxy-2-fluoroglycosides, molecules used in glycoscience as probes, inhibitors, and building blocks for related compounds. The authors focused on using a pyridinium salt as a catalyst to promote the reaction between an O-type glycosyl acceptor and 2-fluoroglycals.
The goal was to achieve efficient formation of these fluorinated sugars while controlling stereochemistry — that is, the three-dimensional arrangement of atoms in the product.
Key findings
The pyridinium-salt catalyst enabled a highly stereoselective addition that produces a diverse collection of 2-fluoroglycosides. The reaction shows excellent preference for syn-addition, meaning the new groups attach on the same face of the sugar-like intermediate.
- Deuterium-labeling experiments indicate that the bond-forming event proceeds in a concerted manner, where bond changes happen in a single coordinated step.
- NMR titrations together with density functional theory (DFT) calculations reveal that the reaction products can inhibit the catalyst, which affects overall efficiency.
- These mechanistic insights also explain why reactions are less efficient when electron-deficient phenols or simple alcoholic substrates are used as acceptors.
Why it matters
2-deoxy-2-fluoroglycosides are important tools and building blocks in chemical biology and the design of bioactive molecules. A stereoselective, catalyst-driven route that provides broad structural access helps researchers assemble these targets more predictably.
Understanding the reaction mechanism and limitations — including product inhibition and reduced performance with certain acceptors — offers a clearer picture of when this approach will work best and where further optimization might be needed.
Disclosure
- Research title: Stereoselective Assembly of 2-Deoxy-2-fluoroglycosides Enabled by Pyridinium Catalysis
- Authors: Shi-Ang Zhai, Zhiwei Ai, Qi Zhang, Qinbo Jiao, Shiping Wang, Chunfa Xu
- Institutions: Fuzhou University, The Fertilizer Institute, Fauji Fertilizer (Pakistan), Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences
- Journal / venue: Organic Letters (2026-01-08)
- DOI: 10.1021/acs.orglett.5c05002
- OpenAlex record: View on OpenAlex
- Links: Landing page
- Disclosure: This post was generated by Artificial Intelligence. The original authors did not write or review this post.